Strong eradication rates with first-
line treatment
NONINFERIORITY ENDPOINT IN PATIENTS WITHOUT AMOXICILLIN- OR CLARITHROMYCIN-RESISTANT STRAINS OF (mITT)
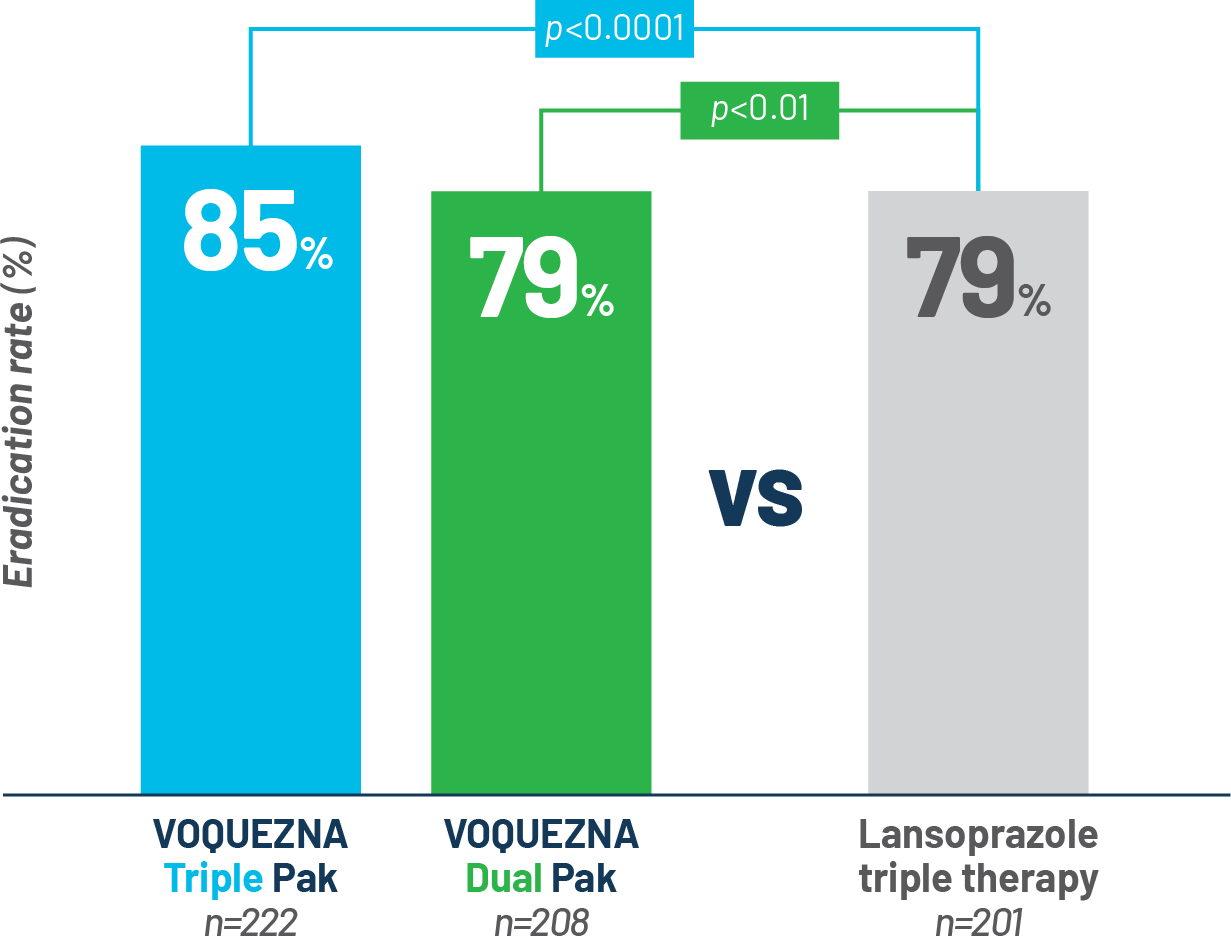
Powerful
eradication rates
Eradication rates in the per-protocol population (PP) were consistent
with the mITT population2
NONINFERIORITY ENDPOINT IN PATIENTS WITHOUT AMOXICILLIN-
OR CLARITHROMYCIN-RESISTANT STRAINS OF (PP)
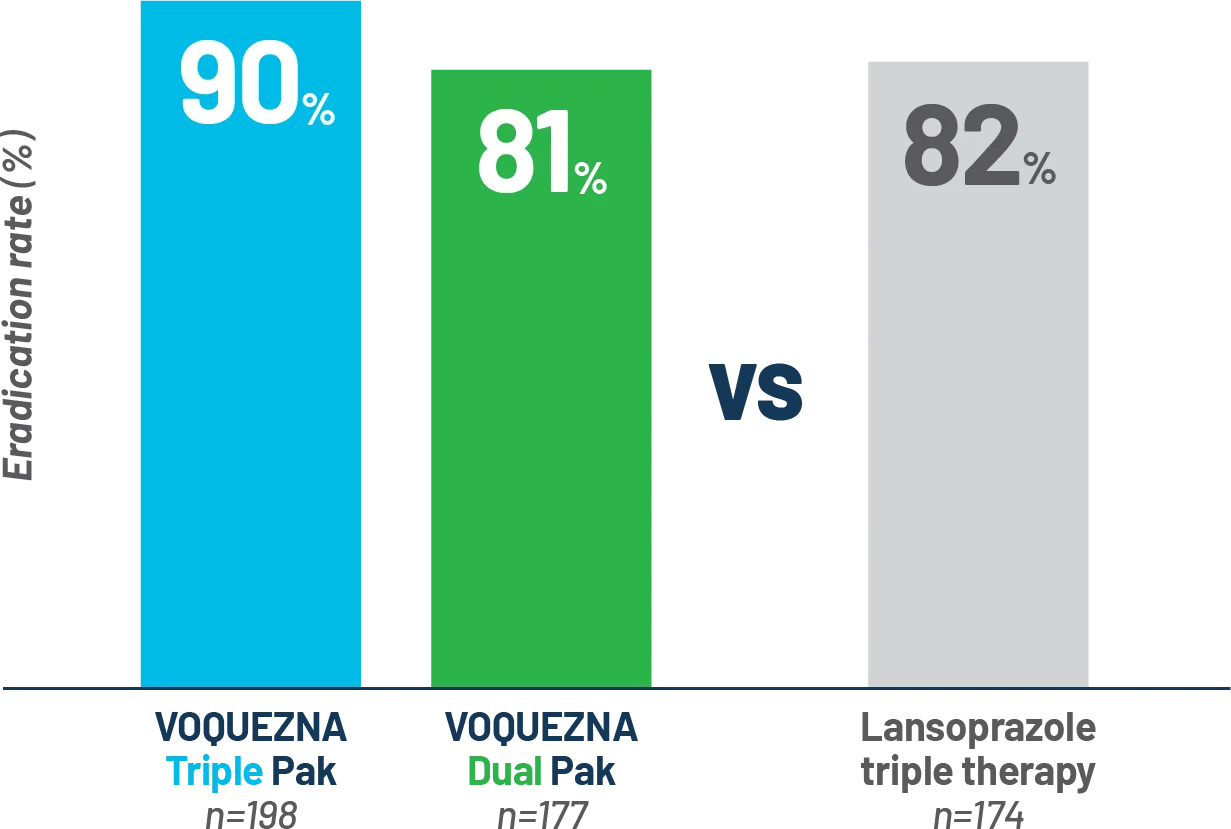
Eradication rate for
VOQUEZNA Triple Pak
in the PP population
was 90%